Introduction
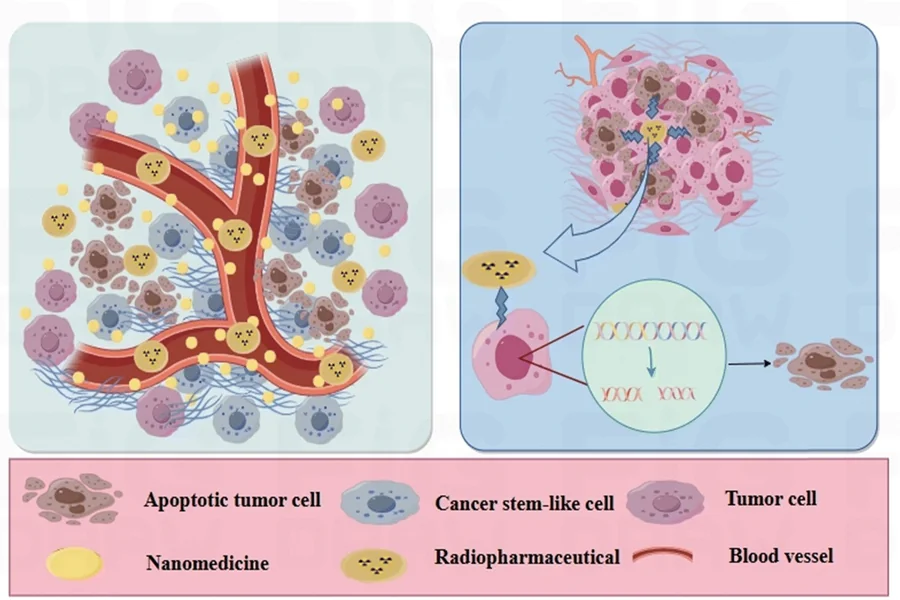
Radioligand therapy (RLT) is an advanced oncological treatment which couples a therapeutic radionuclide, or radioactive isotope, with a cancer-specific cell-targeting module called the ligand. The radioactive isotope, or radionuclide, is a typically unstable atom (specifically defined by the number of protons and neutrons), which emits radiation as it transforms into a more stable form. The ligand selectively binds to biomarkers, receptors, or antigens within the cancer cell, meaning it can be delivered directly to the tumor site. Once this process has occurred, the radionuclide releases cytotoxic radiation, which damages or destroys the cancer cells. This therapeutic discovery has changed the face of cancer treatment, showcasing a significant shift from traditional treatment methods, reducing damage to surrounding healthy tissue, and limiting potential side effects and toxicity. RLT often comes under the bracket of Theranostics, combining molecular targeting and optimized radiation dosimetry, or more broadly, diagnostic and therapeutic approaches. Radiotheranostics confirm the presence of radiolabeled diagnostic biomarkers, identifying viable patients through complex radioligand imaging prior to administration of the radionuclide. While RLT shows ongoing clinical promise, having the potential to drive substantial progress in cancer diagnosis and treatment, it is rapidly becoming one of the most complex and regulated areas of clinical research. Radioligand therapy research has extensive requirements for approval, radiation safety, and optimisation of target strategy, meaning radioligand therapy drug developers have a lot to consider to ensure the safety, efficacy, feasibility, and regulatory compliance of their RLT clinical trials. This article considers emerging radionuclides in RLT, focusing on what developers really need to know.
The current landscape
Food and Drug Administration (FDA) approved and most commonly utilized RLT therapies include Lutathera and Pluvicto, which use a beta-emitting radionuclide known as Lutetium-177 (¹⁷⁷Lu). Following the results of the NETTER-1 trial, the VISION trial, and the PSMAfore trial, applications have been identified across therapeutic areas, specifically neuroendocrine (GEP-NET) and prostate tumors (mCRPC). Other approved radionuclides include Yttrium-90 and Radium-223, for use in liver cancer, lymphoma, and prostate cancer with bone metastases. The success of these RLT therapies has driven exploration into alternative radionuclides, extending research to a range of different tumor types and stages. The global radioligand therapy market, which was valued at around $2.36 billion in 2024, is expected to soar to a staggering $10.91 billion by the end of 2035. Emerging radionuclides may be driving this growth, expanding the therapeutic landscape beyond established isotopes like Lutetium-177, and offering drug developers new opportunities for discovery.
Key emerging radionuclides
β-particle emitters (or beta emitters) are the current standard, including iodine-131, lutetium-177, yttrium-90, strontium-89, and samarium-153, with proven safety and efficacy across clinical research studies. However, key emerging radionuclides for radioligand therapy (RLT) include α-particle-emitters (or alpha emitters) such as Actinium-225 (Ac-225) and Astatine-211 (At-211). While β-particle emitters have relatively low levels of linear energy transfer (LET), α-particle emitters have high LET capabilities, meaning they are capable of depositing a large amount of energy within a short range. This results in highly localised cell damage, aa property first demonstrated in 1994 by Howell et al, who confirmed the relationship between α-particle energy and biological effectiveness in vivo. Targeted α-particle therapy (TAT) could be considered the ‘next wave’ of RLT, gaining momentum throughout early-phase preliminary research, with findings suggestive of potent antitumor activity, durable response rates, and manageable toxicity profiles.
Other promising candidates include:
- Terbium-161 (Tb-161)
- Lead-212 (Pb-212)
- Bismuth-213 (Bi-213)
- Copper-67 (Cu-67)
Ongoing trials will continue to evaluate their safety and efficacy within clinical practice, extending to different disease stages and a broad range of tumor types.
What developers need to consider
While this area of research undergoing a period of rapid growth and innovation is exciting, it also comes with challenges. The infrastructure for the manufacture and distribution of radiopharmaceuticals is still in development, as are the methods for production, purification, and analysis, and regulatory bodies are still optimizing guidelines for this new class of therapy. The production of radionuclides requires strict safety and quality procedures to ensure the stability of the radioactive material, ensuring the safety of both patients and researchers.
RLT drug developers must diversify their radionuclide strategies early, build manufacturing partnerships, and engage regulators proactively. Those who adapt quickly will lead the development of next-generation RLT.
Key factors for R&D teams when assessing new radionuclides:
- Clinical fit – including tumor type, compatibility with targeting vectors, and dosimetry considerations.
- Manufacturing and supply chain – Reactor vs Generator production routes, requirements for novel isotopes.
- Regulatory requirements – Evolving EMA/FDA expectations and documentation/audit requirements for novel radionuclides.
- Integration with theranostics – Pairing therapeutic radionuclides with diagnostic isotopes to enable personalised treatment.
- Cost and commercialisation – Long-term market viability, pricing structure, and reimbursement potential.
Developers who understand both the science and the practicalities will be best positioned to succeed. Isotope production has long lead times, and early planning is essential to secure manufacturing capacity.
Radioligand therapy CRO
Aligning your goals with the expertise and infrastructure of a Radioligand Therapy CRO can enhance the success and commercialisation of your radiopharmaceutical program.
Perceptive Discovery house extensive radiochemistry services and ligand discovery support, being one of the few imaging providers with proven radiolabeling capabilities using ²¹¹At, delivering precision and efficiency for all of your radiochemistry needs.
Resources
Markets & Markets. Radioligand therapy (RLT) market, growth, share, and trends analysis.
The New England Journal of Medicine. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors.
The New England Journal of Medicine. Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer.
The Lancet. Lu-PSMA-617 versus a change of androgen receptor pathway inhibitor therapy for taxane-naive patients with progressive metastatic castration-resistant prostate cancer (PSMAfore): a phase 3, randomised, controlled trial.
BMC Gastroenterology. A prospective, multicenter, open-label, single-arm clinical trial design to evaluate the safety and efficacy of 90Y resin microspheres for the treatment of unresectable HCC: the DOORwaY90 (Duration Of Objective Response with arterial Ytrrium-90) study.
The Lancet. Efficacy and safety of radium-223 dichloride in patients with castration-resistant prostate cancer and symptomatic bone metastases, with or without previous docetaxel use: a prespecified subgroup analysis from the randomised, double-blind, phase 3 ALSYMPCA trial.
Journal of Nuclear Medicine. Clinical Trial Protocol for VIOLET: A Single-Center, Phase I/II Trial Evaluation of Radioligand Treatment in Patients with Metastatic Castration-Resistant Prostate Cancer with [161Tb]Tb-PSMA-I&T.
Lancet Oncology. Trends in Nuclear Medicine and the Radiopharmaceutical Sciences in Oncology: Workforce Challenges and Training in the Age of Theranostics.
Journal of Nucelar Medicine. Lead-212 Radio-DARPin Therapeutic (RDT) targeting delta-like ligand 3 (DLL3) shows promising preclinical antitumor efficacy and tolerability in small cell lung cancer (SCLC).
Pharmaceutics. Bismuth-213 for Targeted Radionuclide Therapy: From Atom to Bedside.
PNAS. Harnessing 64Cu/67Cu for a theranostic approach to pretargeted radioimmunotherapy.
TrAC Trends in Analytical Chemistry. Recent advances in emerging radiopharmaceuticals and the challenges in radiochemistry and analytical chemistry.
Radiation Research. Relative Biological Effectiveness of Alpha-Particle Emitters In Vivo at Low Doses.